|
10/23/2023 0 Comments Pickering emulsion simulation shear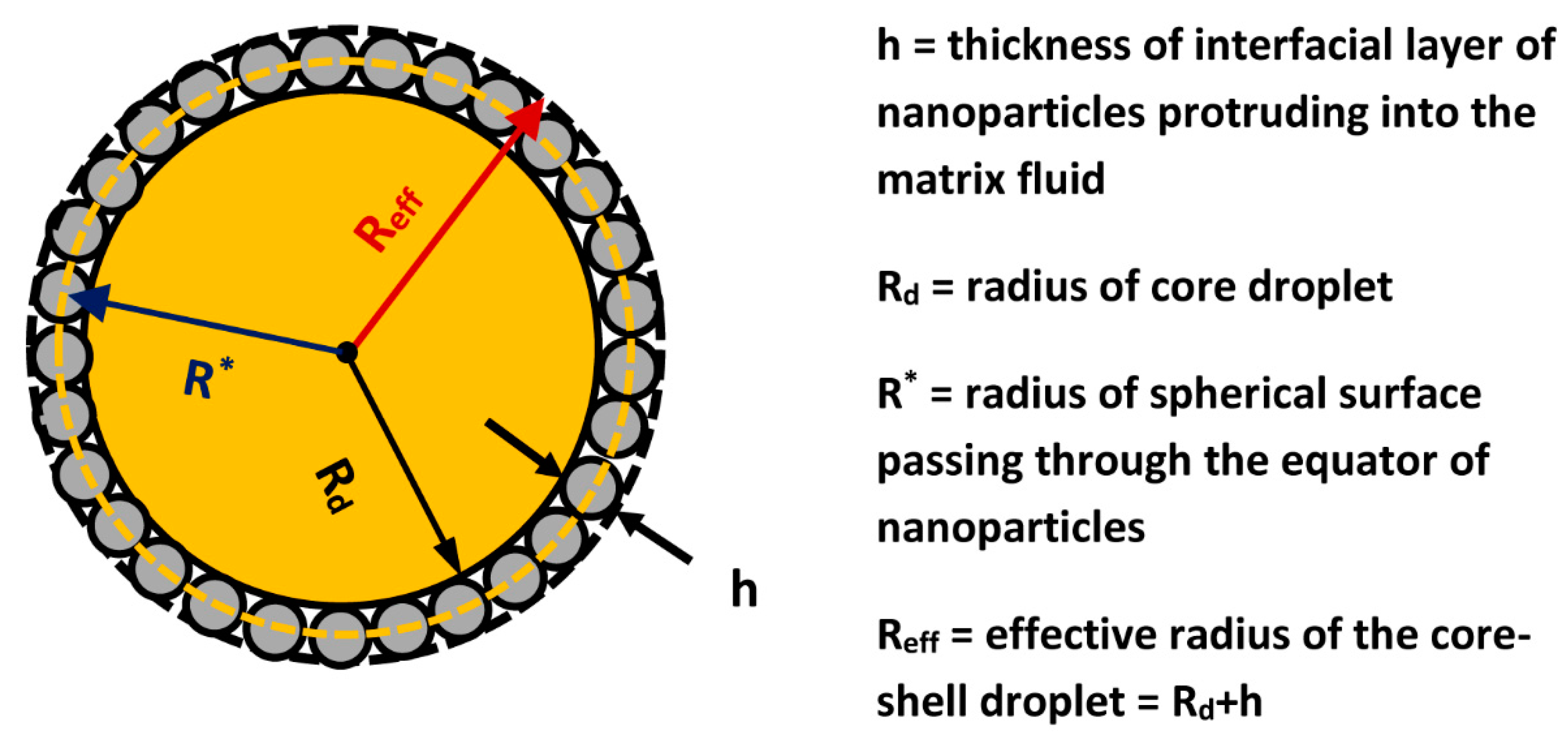
For the A-PS and S-PS/A-PS particle systems, some fully covered droplets were observed in addition to the aggregated domain morphology. In the binary systems, there is no significant partitioning of the particles on the droplet interface, that is to say, the particles are well mixed. All of the larger images, whether a single or binary particle type was used, show the aggregated domain morphology in which the IL droplet is nearly fully covered by the particles. The three images on the left of the figure show the resulting surface coverage of the droplet when a single particle type is used, while the two large images on the right, with additional smaller representative images of selected systems in the middle, show the morphology when two differing particle types are mixed in the emulsion at equal concentrations. Shows the resulting droplet morphologies and partition preferences of the particles, each image showing a IL emulsion droplet suspended in water. The S-PS and AS-PS particles were relatively hydrophobic and negatively charged, while the A-PS particles were relatively hydrophilic and positively charged. For this work, the various surface chemistries of the PS were sulfate (–SO 3H, S-PS, blue color), aldehyde sulfate (–CHO and –SO 3H, AS-PS, green color), or amine (–NH 2, A-PS, green color). In order to observe the effect of particle hydrophobicity and surface charge and learn the resultant particle morphology on the droplets and the partition preference of the particles, 1 μm fluorescent surface-treated polystyrene (PS) particles were employed in conjunction with a confocal laser-scanning microscope. The hydrophobic IL 1-butyl-3-methylimidazolium hexafluorophosphate (), which is immiscible with water, was chosen for the droplet phase. While IL-in-water Pickering emulsions had been created previously, their use of silica nanoparticles prevented easy observation of the resulting particle morphology, and thus, fluorescent micron-sized particles were used in this work. Our first foray into ionic liquid-based Pickering emulsions was to simply replace the droplet phase in a conventional oil-in-water emulsion with an IL to create an IL-in-water Pickering emulsion. Deviating from the conventional oil-in-water emulsion systems, employing ILs allows for interesting interfacial phenomena due to the charged nature of the IL and other factors intrinsic to the liquid including ion ordering, interfacial tension, etc. Surfactant-free, solid-stabilized Pickering emulsions can act as templates for these studies in which the ionic liquid subsists as the droplet phase, continuous phase, or both phases in the emulsion. These suggest unique applications in extraction, catalysis, reaction schemes, etc., and provide even more extensive tunability through the complex interactions of differing choices of particles and ionic liquids. Additionally, by incorporating particles into these IL-liquid systems, novel and varied behaviors can be achieved. The extremely high tunability of the material properties of ionic liquids (ILs), caused by the virtually endless combinations of anions and cations, allows ILs to be unique candidates for the study of liquid-liquid interfaces. Unique morphologies of aqueous and nonaqueous ionic liquid Pickering emulsions There continues to be active research in Pickering emulsions, immiscible phases, and ILs, and one particularly relevant application is employing ionic liquid-in-water emulsions as “a new class of fluorescent sensors for metal ions”. Illustrates some of these phenomena-particle self-assembly on an emulsion surface, particle “bridging” between emulsion droplets, and spontaneous transport across the liquid-liquid interface-as well as an example of the molecular dynamics (MD) simulations used to further explore the fundamentals of IL interactions with other phases. It explores ionic liquid-liquid interfaces and their role in ionic liquid Pickering emulsions through both experimental and simulation approaches, including several unique and fascinating interface phenomena. This work focuses in particular on systems involving solid particles and immiscible liquid phases. Ionic liquids’ complex interactions with other materials enable multiphase systems that are both theoretically fascinating and potentially useful. The unique properties and rich variety of ionic liquids (ILs) make them promising for a wide range of applications and the potential of pure ILs is multiplied by introducing them to multicomponent systems.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
